Flytzani-Stephanopoulos's research group has been active in designing catalysts with a reduced amount of precious metals used to generate high-grade hydrogen for fuel cells. The water-gas shift reaction, in which carbon monoxide is removed from the fuel cell by reacting with water to produce carbon dioxide and hydrogen, is a key step in fuel processing hydrogen. Catalysts, such as metal oxides prepared with precious metals like platinum, are used to lower the reaction temperature and increase the production of hydrogen.
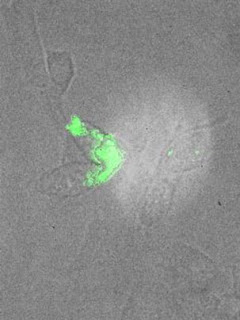
The Tufts group was first to demonstrate that catalysts prepared with atomic-level dispersions of gold or platinum in oxide supports, such as cerium oxide or iron oxide, show the highest activity for the water-gas shift reaction. Adding more metal to form nanoparticles on the support oxide does not improve the activity; it simply wastes the precious metal. Highly active and stable catalysts are required for integration in fuel cell systems. To improve the stability of the precious metal with temperature and length of operating time, the Tufts group was first to show that adding small amounts of gaseous oxygen could have the desired structural effect of keeping gold fully dispersed and available to react.
Professor Stephanopoulos and her research group are presently investigating ways to further improve the activity of doped metal oxides by identifying which oxide structure binds gold stronger. The researchers synthesize single crystal oxides at the nanoscale with specific shapes (rods, cubes, polyhedra) and crystal faces; dope these with various metals and study them as catalysts for alcohol steam reforming, the water-gas shift reaction, hydrocarbon oxidation and other reactions, including sulfur removal reactions. The aim is to benefit fuel processing overall at all scales—from providing power to a laptop or cell phone to increasing energy from a power plant—and for any fuel gas stream, including coal-derived gas.
"There are always new things to discover in materials science which impact catalysis," said Flytzani-Stephanopoulos. "With new methods for the synthesis of nanomaterials rich in defects, and with proper doping of these structures with a variety of metals, it's like starting from scratch again to understand their chemistry and tailor their catalytic properties to optimize both their activity and selectivity for a large number of reactions of interest to clean and sustainable energy production."
This year 486 members were selected as Fellows because of their scientifically or socially distinguished efforts to advance science or its applications. This year's AAAS Fellows will be announced in the AAAS News & Notes section of the journal Science on 19 December 2008. New Fellows will be honored during the 2009 AAAS Annual Meeting in Chicago on February 14. ###
The American Association for the Advancement of Science (AAAS) is the world's largest general scientific society. AAAS was founded in 1848, and includes some 262 affiliated societies and academies of science, serving 10 million individuals. The non-profit AAAS (www.aaas.org) is open to all and fulfills its mission to "advance science and serve society" through initiatives in science policy; international programs; science education; and more.
Tufts University, located on three Massachusetts campuses in Boston, Medford/Somerville, and Grafton, and in Talloires, France, is recognized among the premier research universities in the United States. Tufts enjoys a global reputation for academic excellence and for the preparation of students as leaders in a wide range of professions. A growing number of innovative teaching and research initiatives span all Tufts campuses, and collaboration among the faculty and students in the undergraduate, graduate and professional programs across the university's schools is widely encouraged.
Contact: Alex Reid Alexander.reid@tufts.edu 617-627-4173 Tufts University





























 " id="BLOGGER_PHOTO_ID_5291650074890931618" border="0">
" id="BLOGGER_PHOTO_ID_5291650074890931618" border="0">

