Researchers turn everyday paper into resilient, rechargeable energy storage device
Troy, N.Y. – Researchers at Rensselaer Polytechnic Institute have developed a new energy storage device that easily could be mistaken for a simple sheet of black paper.
 | The nanoengineered battery is lightweight, ultra thin, completely flexible, and geared toward meeting the trickiest design and energy requirements of tomorrow’s gadgets, |
implantable medical equipment, and transportation vehicles.
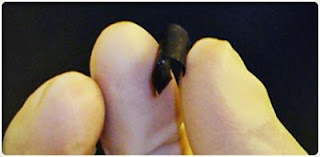 | Along with its ability to function in temperatures up to 300 degrees Fahrenheit and down to 100 below zero, the device is completely integrated and can be printed like paper. |
The device is also unique in that it can function as both a high-energy battery and a high-power supercapacitor, which are generally separate components in most electrical systems. Another key feature is the capability to use human blood or sweat to help power the battery.
Details of the project are outlined in the paper “Flexible Energy Storage Devices Based on Nanocomposite Paper” published Aug. 13 in the Proceedings of the National Academy of Sciences.
The semblance to paper is no accident: more than 90 percent of the device is made up of cellulose, the same plant cells used in newsprint, loose leaf, lunch bags, and nearly every other type of paper.
Rensselaer researchers infused this paper with aligned carbon nanotubes, which give the device its black color. The nanotubes act as electrodes and allow the storage devices to conduct electricity. The device, engineered to function as both a lithium-ion battery and a supercapacitor, can provide the long, steady power output comparable to a conventional battery, as well as a supercapacitor’s quick burst of high energy.
The device can be rolled, twisted, folded, or cut into any number of shapes with no loss of mechanical integrity or efficiency. The paper batteries can also be stacked, like a ream of printer paper, to boost the total power output.
“It’s essentially a regular piece of paper, but it’s made in a very intelligent way,” said paper co-author Robert Linhardt, the Ann and John H. Broadbent Senior Constellation Professor of Biocatalysis and Metabolic Engineering at Rensselaer.
“We’re not putting pieces together – it’s a single, integrated device,” he said. “The components are molecularly attached to each other: the carbon nanotube print is embedded in the paper, and the electrolyte is soaked into the paper. The end result is a device that looks, feels, and weighs the same as paper.”
The creation of this unique nanocomposite paper drew from a diverse pool of disciplines, requiring expertise in materials science, energy storage, and chemistry. Along with Linhardt, authors of the paper include Pulickel M. Ajayan, professor of materials science and engineering, and Omkaram Nalamasu, professor of chemistry with a joint appointment in materials science and engineering. Senior research specialist Victor Pushparaj, along with postdoctoral research associates Shaijumon M. Manikoth, Ashavani Kumar, and Saravanababu Murugesan, were co-authors and lead researchers of the project. Other co-authors include research associate Lijie Ci and Rensselaer Nanotechnology Center Laboratory Manager Robert Vajtai.
The researchers used ionic liquid, essentially a liquid salt, as the battery’s electrolyte. It’s important to note that ionic liquid contains no water, which means there’s nothing in the batteries to freeze or evaporate. “This lack of water allows the paper energy storage devices to withstand extreme temperatures,” Kumar said.
Along with use in small handheld electronics, the paper batteries’ light weight could make them ideal for use in automobiles, aircraft, and even boats. The paper also could be molded into different shapes, such as a car door, which would enable important new engineering innovations.
“Plus, because of the high paper content and lack of toxic chemicals, it’s environmentally safe,” Shaijumon said.
Paper is also extremely biocompatible and these new hybrid battery/supercapcitors have potential as power supplies for devices implanted in the body. The team printed paper batteries without adding any electrolytes, and demonstrated that naturally occurring electrolytes in human sweat, blood, and urine can be used to activate the battery device.
“It’s a way to power a small device such as a pacemaker without introducing any harsh chemicals – such as the kind that are typically found in batteries – into the body,” Pushparaj said.
The materials required to create the paper batteries are inexpensive, Murugesan said, but the team has not yet developed a way to inexpensively mass produce the devices. The end goal is to print the paper using a roll-to-roll system similar to how newspapers are printed.
“When we get this technology down, we’ll basically have the ability to print batteries and print supercapacitors,” Ajayan said. “We see this as a technology that’s just right for the current energy market, as well as the electronics industry, which is always looking for smaller, lighter power sources. Our device could make its way into any number of different applications.”
The team of researchers has already filed a patent protecting the invention. They are now working on ways to boost the efficiency of the batteries and supercapacitors, and investigating different manufacturing techniques.
"Energy storage is an area that can be addressed by nanomanufacturing technologies and our truly inter-disciplinary collaborative activity that brings together advances and expertise in nanotechnology, room-temperature ionic liquids, and energy storage devices in a creative way to devise novel battery and supercapacitor devices," Nalamasu said. ###
The paper energy storage device project was supported by the New York State Office of Science, Technology, and Academic Research (NYSTAR), as well as the National Science Foundation (NSF) through the Nanoscale Science and Engineering Center at Rensselaer.
About Rensselaer: Rensselaer Polytechnic Institute, founded in 1824, is the nation’s oldest technological university. The university offers bachelor’s, master’s, and doctoral degrees in engineering, the sciences, information technology, architecture, management, and the humanities and social sciences. Institute programs serve undergraduates, graduate students, and working professionals around the world.
Rensselaer faculty are known for pre-eminence in research conducted in a wide range of fields, with particular emphasis in biotechnology, nanotechnology, information technology, and the media arts and technology. The Institute is well known for its success in the transfer of technology from the laboratory to the marketplace so that new discoveries and inventions benefit human life, protect the environment, and strengthen economic development.
Contact: Michael Mullaney
mullam@rpi.edu 518-276-6161
Rensselaer Polytechnic InstituteTechnorati Tags:
Nano or
Nanotechnology and
Nanotech and
batteries or
Rensselaer Polytechnic Institute and
Nanocomposite Paper or
National Intelligence Estimate Prospects for Iraq’s Stability FULL TEXT and
The Leaning Tower, Pisa, Italy and
Scientists train nano-'building blocks' to take on new shapesThe Virtuous Republic,
Is It Just Me?,
Rosemary's Thoughts,
DeMediacratic Nation,
Adam's Blog,
Big Dog's Weblog,
Shadowscope,
Webloggin,
The Bullwinkle Blog,
Phastidio.net,
Leaning Straight Up,
,
third world county,
Faultline USA,
Woman Honor Thyself,
The Crazy Rants of Samantha Burns,
The Uncooperative Radio Show!,
The World According to Carl,
Walls of the City,
Pirate's Cove,
Blue Star Chronicles,
The Pink Flamingo,
CommonSenseAmerica,
Dumb Ox Daily News,
The Yankee Sailor, and
Church and State, thanks to
Linkfest Haven Deluxe.
Trackback URL: http://haloscan.com/tb/sookietex3/8221617620114451105
13 Trackbacks to this Post
Joe Kaufman of Americans Against Hate - on H Excerpt: Joe Kaufman, Americans Against Hate and CAIR Watch, appears on Hannity Colmes to discuss the NYC madrassa and the upcoming NYC islamist day parade. Colmes plays the typical dhimmi.Trackposted to Outside the Beltway, Blog @ MoreWhat.com, Perri Nelson's... Weblog: The Amboy Times Tracked: 08.25.07 - 5:34 pm
Ah, sweet Saturday; OTB Excerpt: What have I accomplished today? Let's see...I went through emails, I worked on my open trackbacks, I got a new mouse that had a cord that was too short (actually, my brother got it for me because I don't leave the apartment, but that's a whole di ..... Weblog: Rosemary's Thoughts Tracked: 08.25.07 - 6:02 pm
Cate Blanchett is to Play Elizabeth I Once Again Excerpt: If you are going to make a movie about the life of Queen Elizabeth I, who else could you get to play the role of the Tudor Queen but Cate Blanchett? No one else could do it, of course. Not after her Oscar winning performance as the Queen in Elizabeth... Weblog: Adeline and Hazel Tracked: 08.25.07 - 6:05 pm
Women who smell bad Excerpt: Once you bathe them all women of the same race smell the same Weblog: Planck's Constant Tracked: 08.25.07 - 7:25 pm
Shadowscope Immigration Activists Protest Deportation of Arellano… According to the article from Newstext hundreds of Immigration activists marched in Los Angeles yesterday in order to show solidarity with Elvira Arellano, the woman that was deported last week after having hidden in a Chicago church for a year…. 08.26.07 08:31 am
walls of the city Says: premature … somethingoranother
So I have a small confession to make. I was going to be a good little boy, and postpone any major firearm-related purchases until Tuesday’s National Exercise Your Rights Day - after all, it would make the largest impact then…. August 26th, 2007 at 11:04 am
Hangings, Executions and other good stuff Excerpt: Iran has hanged up to 30 people in the past month amid a clampdown prompted by alleged U.S.-backed plots to topple the regime, while the EU urges Texas to end executions. Iran: (here via here) Many executions have been carried Weblog: Right Truth Tracked: 08.26.07 - 12:39 pm
When Fake News is Presented as Truth by the Main S Excerpt: Its a mystery to me as to why the Old Media insists on reporting the amateurish propaganda that comes out of the middle east while ignoring reporting any news stories that promote the best interest of the United States. Its a very strange phenomena t... Weblog: Blue Star Chronicles Tracked: 08.26.07 - 4:46 pm
Americans, Iraqis interact at historical monument Excerpt: It has been more than 10 years since any Iraqi native has been allowed to visit the Ziggurat of Ur, which is the most dominant landmark on Ali Base, because during the reign of Suddam Hussein the installation was used by the Iraqi army.Weblog: Rosemary's Thoughts Tracked: 08.26.07 - 7:13pm
Adam's Blog The Trouble With Huckabee… Mike Huckabee is a decent man, but his fiscal record dooms his ability to bring Conservatives together. Also, Howard Dean and the DNC doing something right for a change, the Deficit’s falling but a Democrat tax and spending increase could make i… 08.26.07 - 11:08pm
Adeline and Hazel Owen Wilson Hospitalized Following Possible Suicid… The National Enquirer later revealed that he had been found by a family member after he had sliced his left wrist and taken an undetermined number of pills. His condition was listed as very serious and his prognosis grim. He has sense been upgraded a….. 08.27.07 1.00am
Right Voices Hey MSM You Forgot To Report That France’s Health Care System Broken and Should Copy US!… French Expert Details Failures in System, Compares to U.S. In a nutshell, the system is not sustainable anymore. “It’s true we really have good access, but what if the system is not sustainable anymore?” says Teil. “It’s … 08.27.07 1:21pm
Leaning Straight Up Says: More cartoon “Dhimmitude”: Newspapers refuse to print Opus cartoon Mocking Islam I have avoided the use of this term in the past as it is pejorative and far too convenient for my tastes. But more and more, the ass kissing of Muslim hyper-sensitivity has been irking me, so I see no reason to continue ignoring it for what it is. August 27th, 2007 at 4:57 pm


















